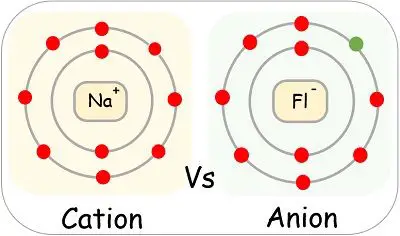
We can divide the ions into two categories: cation and anion, based on the charge they bear. However, they both get generated from a neutral atom.
The cations consist of more positively charged protons than that of negative electrons. And due to this extra positivity, they have a positive charge. In contrast, the anions are negatively charged as they possess more electrons than protons.
The cations being positively charged, tend to move towards the negative electrode. Whereas, the positive electrodes attract the negatively charged anion.
Most of the time, the metals are eager to donate the valence electrons, so as to stabilize their octet. Thus, they are considered electropositive. And have a tendency to form a cation.
Oppositely, the non-metals are electronegative. And thus, they accept the electrons to fulfil their octet. Therefore, they tend to make anion.
In this context, we will learn the primary differences between the cation and anion in detail.
Content: Cation Vs Anion
- Comparison Chart
- What are Ions? And how are they formed?
- What are Cations?
- What are Anions?
- Key Differences
- Conclusion
Comparison Chart
| Basis for Comparison | Cation | Anion |
|---|---|---|
| Meaning | An atom or molecule having more number of protons than electrons and are positively charged is called cation. | An atom or molecule having more number of electrons than protons and are negatively charged is called anion. |
| Charge | These are positively charged. | These are negatively charged. |
| Formed of | Cation are formed from metal atoms. | Anion are formed from non-metals atoms. |
| Compounds formed | Cation combine with the anions to form the ionic bonds. | Anion combine with the cations to form the ionic bonds. |
| Electrolysis | Cation always move towards the cathode, which produces the negative charge. | Anion always move towards the anode, which produces the positive charge. |
| Examples | Iron (Fe2+), Sodium (Na+), Lead (Pb2+). | Fluoride (F-), Bromide (Br-), Iodide (I-), Nitride (N3-) and Hydride (H-). |
What are Ions? And how are they formed?
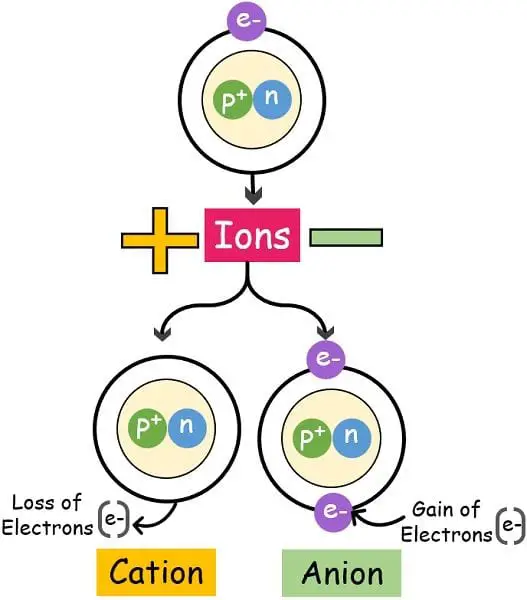
We can define ions as “A charged particles due to an unequal number of protons and electrons.
Our earth comprises several different elements exiting in various forms. However, these elements aren’t stable much in their atomic state. The atoms are neutral bodies with no charges. In order to stabilize themselves and match the stability of noble elements, they transform into an ionic state.
The atom either gives out or takes in the electron to become an ion. Moreover, the negative and positive charges generated have attraction between them. This leads to the formation of compounds with ionic bonds.
What are Cations?
The ions with permanent positive charges are cations. They have more positive protons than that of negative electrons. Thus the overall charge of the atom becomes positive. We refer to this as a cationic atom.
Every atom in this universe wants to stabilize. In order to do so, the electropositive atoms donate their valence electrons. In this way, they balance and match E.C. of the nearest noble gas.
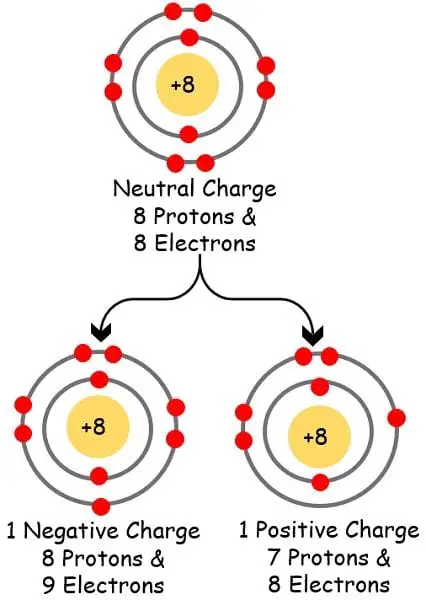
The atom removes the electrons with a lower affinity. Due to this removal of electrons number of electrons reduces thereby decreasing the negative charge. In contrast, the number of protons rises which elevates the positive charge of the atom.
As soon as this happens, the neutral atom transforms into a cation.
An electropositive atom will search for a receiver that is devoid of electrons and will donate its electrons to it. Generally, this character is present in metals. They tend to give electrons and gain a positive charge.
We denote the cations by the (+) symbol on the top right of the element.
For example: Mg2+, Na+, Ca2+ etc.
The negative electrode attracts the cations towards itself as the electricity passes during the electrolysis reaction.
Properties of Cation
- The cations are positively charged.
- They bear more positive protons than that of electrons.
- The metals have the tendency to form cations as they are highly electropositive.
- They react with anions through ionic bonds and produce ionic compounds.
- During the electrolysis process, cations tend to displace towards the negative electrode.
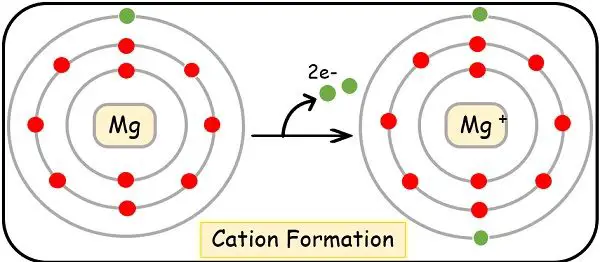
Formation of Cations
We will learn the formation of the cation with the help of an example.
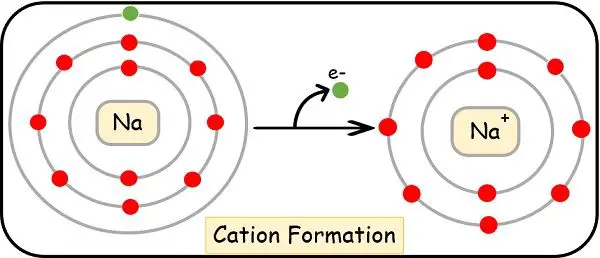
Let’s take the example of sodium having the electronic configuration as:
11Na = [Ne], 1s2, 2s2, 2p6, 3s1
In order to stabilize its octet, the sodium atom gives away the 1 electron of its valence shell. As the atom has a lower affinity to that electron, it readily detaches and moves to the electronegative one.
As soon as this happens, the sodium remains with 10 electrons and 11 protons. Due to the higher number of positive charges, the sodium will become cation. Now its E.C. will change as:
Na+= (Ne), 1s2, 2s2, 2p6
Another example is magnesium with E.C. as:
12Mg = (Ne),1s2, 2s2, 2p6, 3s2
It is also an electropositive element. And thus, it donates two of its valence shell electrons to become a cation.
After becoming a cation its E.C. will change as:
Mg2+= (Ne), 1s2, 2s2, 2p6
Some more examples of Cations
- Sodium-ion (Na+)
- Potassium ion (K+)
- Silver ion (Ag+)
- Hydronium (H3O+)
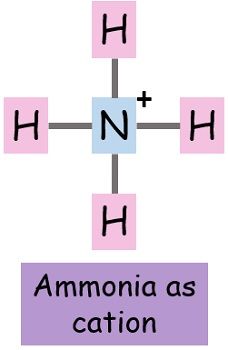
- Ammonium (NH4+)
What are Anions?
The ions that possess a negative charge are anions. In cases, where the atom is trying to stabilize itself by gaining electrons to complete the octet.
A neutral atom becomes an anion when it attracts one or two electrons to its valence orbit. Due to the gain of extra electrons, the number of negative charges increases. In contrast, the number of protons becomes less thereby lowering the positive charge.
Due to the elevation in the negative charge, the neutral atom transforms to anion.
In order to gain the electrons, there should be a donor who is willing to donate the electrons. If the donor atom will have a lesser affinity towards some electrons, it will pass these electrons to others. And will become stable.
The elements which tend to produce an anion are referred to as electronegative elements. This is because they always have a scope to add electrons in the valence orbit to attain stability. Non-metals being electronegative readily produce anions.
The gained negative charge is depicted with a (-) symbol written over the top right of the element.
For example, Cl–, Fl–, etc.
anions displace towards the positively charges electrode.
Properties of Anions
- The anions are negatively charged.
- Anions possess a higher number of electrons than that of protons.
- Due to the higher electron count, they have a permanent negative charge.
- The positive electrode attracts the negative anions during the process of electrolysis.
- These anions react to cations through ionic bonds and generate the ionic compound.
- The non-metals produce anions in most cases as they are highly electronegative.
Note: It is not always necessary that the non-metals form anion only. There are certain exceptions like NH4+. It has no metallic element but still, it is cationic.
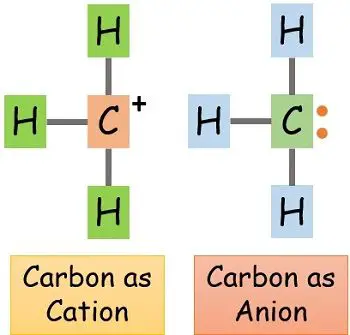
Similarly, carbon which is considered a non-metal can serve both the role of cation and anion as the requirement.
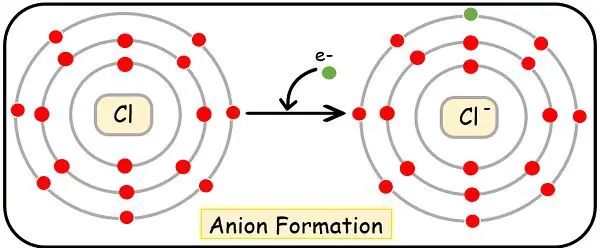
Formation of Anion
We will understand the formation of the anion with the help of examples.
Let’s take the example of chlorine with Electronic configuration as:
17Cl = [Ne], 3s2, 3p5
It is a non-metal with an electronegative tendency. Thereby is ready to attract 1 electron to achieve the nearest noble gas configuration. It possesses 7 electrons in its outermost shell, thus it needs three electrons to stabilize its octet.
As soon as, it attracts the 3 electrons to the valence orbit, it will transform into an anion. This is chloride ion with E.C. as:
(Cl)–= [Ne], 3s2, 3p6
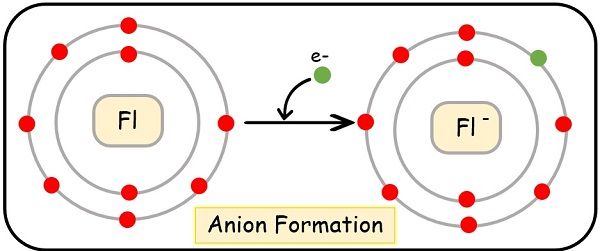
Another example can be seen of fluorine. It is also a non-metal with electronic configuration:
9Fl = [He], 2s2 2p5
Here, also the fluorine requires only one electron to fulfil its valence orbit and gain the E.C. of the nearest noble gas.
(F)–= [He], 2s2, 2p6
Some more examples of Anions
- Hydride (H–)
- Nitride (N3–)
- Bromide (Br–)
- Fluoride (F–)
- Chloride (Cl–)
Key Differences Between Cation and Anion
- The cations are positively charged ions while the anions are negatively charged ions.
- To form a cation, a neutral atom has to donate its electrons. But for the anion formation, the atom gains the electrons.
- Cations attain stability by losing the electrons while the anions complete their octet by gaining the electrons.
- The cations are often smaller in size but the anions are usually larger.
- In cations number of protons are more while that of electrons is less. In contrast, anions have a higher number of electrons and a lower number of protons.
- During the process of electrolysis, the negative electrode attracts the anions. On the other hand, during the same process, the positive electrode attracts the anions.
- Electropositive elements like metals form the cations. Whereas, electronegative elements like non-metals tend to produce an anion.
Conclusion
Cation and anion are among the common words of chemistry. But if we go in-depth, there are many other similar terms too, like neutrons, protons, electrons. These are the subatomic particles of an atom, which is the most tin particle and is invisible to the naked eyes. Ions are formed of such atoms or groups of atoms.
As we discussed above that the ion tends to be stable if it has an equal number of protons and electrons, but when it loses or gains the protons or electrons, it gains the positive or negative charge and is said as cations or anions. In this article, we discussed the ions differentiated by the net charge held by them.

Leave a Reply